Treatment - Cancer Currents Blog
Cancer treatment related news, with context from leading experts. Includes articles on new therapies, treatment side effects, and important trends in treatment-related research.
-
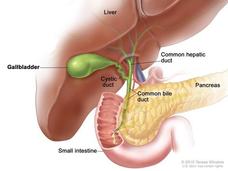
Durvalumab Modestly Improves Survival in Advanced Biliary Tract Cancer
Adding durvalumab (Imfinzi) to standard chemotherapy modestly extended how long people with advanced biliary tract cancer lived, results from the TOPAZ-1 trial show. The immunotherapy drug may now be the standard first-line therapy for this hard-to-treat cancer.
-
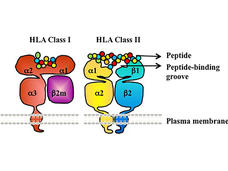
Cancer Immunotherapies Don’t Work for Everyone: HLA Gene May Explain Why
A specific form of the HLA gene, HLA-A*03, may make immune checkpoint inhibitors less effective for some people with cancer, according to an NCI-led study. If additional studies confirm the finding, it could help guide the use of these commonly used drugs.
-
Can mRNA Vaccines Help Treat Cancer?
The success of mRNA vaccines for COVID-19 could help accelerate research on using mRNA vaccine technology to treat cancer, including the development of personalized cancer vaccines.
-
Should CAR T Cells Be Used Earlier in People with Non-Hodgkin Lymphoma?
Results from three clinical trials have found that CAR T cells may be superior to standard treatments for patients with B-cell NHL that has not responded to treatment (refractory) or has returned after treatment (relapsed).
-
When Ovarian Cancer Returns, Surgery May Be a Good Choice for Selected Patients
For patients with recurrent ovarian cancer who meet strict criteria, additional surgery may improve survival, results from a large clinical trial show.
-
Immunotherapy Combination Most Effective as Initial Treatment for BRAF+ Melanoma
Clinical trial finds that ipilimumab (Yervoy) and nivolumab (Opdivo) combo is superior to a combination of the targeted therapies dabrafenib (Tafinlar) and trametinib (Mekinist) as the first treatment for metastatic BRAF-positive melanoma.
-
Shorter, More Intensive Radiation Safe after Surgery for Prostate Cancer
Many with prostate cancer can safely receive shorter, higher-dose radiation therapy after surgery, a new study has found. The approach, called HYPORT, didn’t harm patients’ quality of life compared with the standard radiation approach, trial finds.
-
Tecartus Becomes First CAR T-Cell Therapy Approved for Adults with ALL
The CAR T-cell therapy Tecartus has become the first such treatment approved by FDA to treat adults with acute lymphoblastic leukemia (ALL). The approval is for patients whose cancer has not responded to treatment or returned after treatment.
-
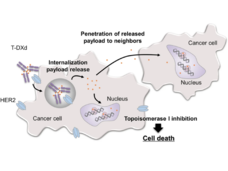
Enhertu May Be Preferred Therapy for Some Metastatic Breast Cancers
In people with metastatic HER2-positive breast cancer, the targeted drug trastuzumab deruxtecan (Enhertu) markedly lengthened progression-free survival compared with trastuzumab emtansine (Kadcycla), new study results show.
-
Pembrolizumab May Help Prevent Early-Stage Melanoma from Returning
Trial results show patients who received the immunotherapy pembrolizumab (Keytruda) after surgery to remove high-risk stage II melanomas were less likely to have the cancer come back than those who received no treatment after surgery.
-
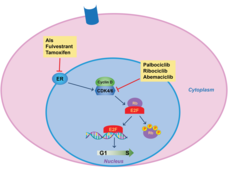
Combo of Ribociclib, Letrozole Improves Survival in Advanced Breast Cancer
In a large clinical trial, women with HR-positive, HER2-negative metastatic breast cancer treated with ribociclib (Kisqali) and letrozole (Femara) as their initial treatment lived approximately 1 year longer than women treated with letrozole only.
-
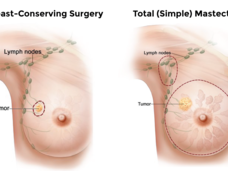
Breast Cancer Surgery Choice May Affect Young Survivors’ Quality of Life
Women with early-stage breast cancer who had one or both breasts surgically removed (a unilateral or bilateral mastectomy) had lower scores on a quality-of-life survey than women who had breast-conserving surgery, a new study has found.
-
FDA Approval of Rylaze Will Address Drug Shortage for Childhood ALL
FDA has approved a new form of asparaginase called Rylaze. The drug was developed to help alleviate shortages of Erwinia asparaginase, a key part of treatment for children and adults with acute lymphoblastic leukemia.
-
For Kids with Medulloblastoma, Trial Suggests Radiation Can Be Tailored
Standard radiation for medulloblastoma can cause long-term damage to a child’s developing brain. A new clinical trial suggests that the volume and dose of radiation could be safely tailored based on genetic features in the patient’s tumor.
-
For Advanced Prostate Cancer, Radiopharmaceutical Improves Survival
A drug called Lu177-PSMA-617 may be a new option for treating advanced prostate cancer. In a large clinical trial, adding the drug—a type of radiopharmaceutical—to standard treatments improved how long participants lived.
-
FDA Approval of KRAS Inhibitor Sotorasib for Lung Cancer Hailed as Milestone
FDA has approved the first KRAS-blocking drug, sotorasib (Lumakras). The approval, which covers the use of sotorasib to treat some patients with advanced lung cancer, sets the stage for other KRAS inhibitors already in development, researchers said.
-
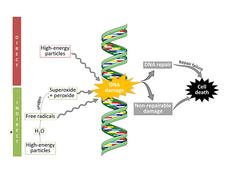
Avasopasem Shields Normal Cells from Radiation, Helps Kill Cancer Cells
A drug called avasopasem manganese, which has been found to protect normal tissues from radiation therapy, can also make cancer cells more vulnerable to radiation treatment, a new study in mice suggests.
-
Nivolumab and Relatlimab Combination Shows Promise in Advanced Melanoma
People with advanced melanoma treated with two immunotherapy drugs—nivolumab (Opdivo) and a new drug called relatlimab—lived longer without their cancer getting worse than those treated only with nivolumab, results from a large clinical trial show.
-
Nivolumab-Based Combinations Improve Survival in Advanced Esophageal Cancer
A treatment regimen that combines the immunotherapy drug nivolumab (Opdivo) with either another immunotherapy drug or chemotherapy may be a new initial treatment option for people with advanced esophageal cancer, a large clinical trial finds.
-
For Hairy Cell Leukemia, Drug Combination Leads to Long-Lasting Remissions
In a small study, vemurafenib (Zelboraf) and rituximab (Rituxan) helped 85% of participants stay in remission for nearly 3 years. The study involved 30 people with hairy cell leukemia that had come back after or had not responded to previous treatment.