What is NCI’s current fiscal year 2026 (FY26) budget?
The Consolidated Appropriations Act, 2026 (H.R. 1748) provides NCI a total appropriation of $7.35 billion, an increase of $128 million compared to the FY 2025 budget.
Where does NCI’s funding come from?
As a federal agency, NCI receives its budget from the United States Congress. The President submits a budget request and spending priorities to Congress, but only Congress can actually provide funding—whether mandatory (direct spending typically dictated by statute, not appropriations bills) or discretionary funding, which is provided via the annual appropriations process. Appropriations are decisions made by Congress about how to designate discretionary federal spending. Congress provides the amount of funds that may be spent, along with guidance regarding funds available for certain initiatives (e.g., Childhood Cancer Data Initiative), and the period of time when NCI can use funds. The federal government's fiscal year (FY) runs from October 1 to September 30.
What steps are involved in the budget process?
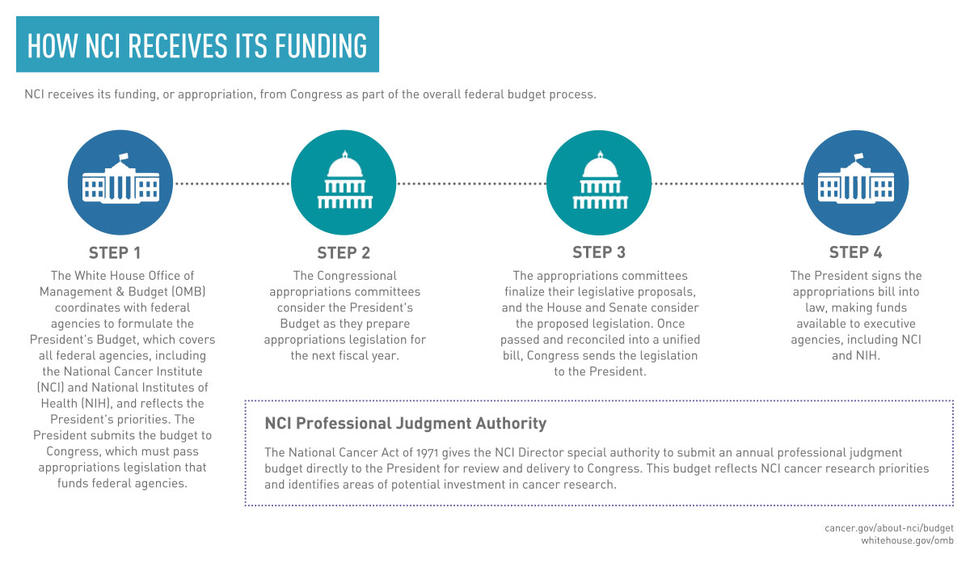
The graphic below outlines each of the major steps in NCI’s funding process.
The federal budget process involves the preparation of separate reports and requests that relate to each other in varying degrees.
Each year NCI prepares and submits the NCI Annual Plan and Professional Judgment Budget directly to the President and Congress for review. The Annual Plan and Professional Judgment Budget is unique to NCI, and this process is required by the National Cancer Act of 1971. The Annual Plan and Professional Judgment Budget identifies NCI’s research priorities and provides a high-level overview of optimum funding needed to advance cancer research. It’s typically published over a year in advance of the fiscal year it outlines.
The White House’s Office of Management and Budget prepares the President’s Budget Proposal in coordination with all federal agencies. This annual document requests funding levels for the federal government for the upcoming fiscal year according to the President’s priorities. The President submits the budget request for Congress to consider when passing appropriations legislation to fund federal agencies.
NCI also prepares the Congressional Justification, in coordination with NIH, the Department of Health and Human Services, and the Office of Management and Budget. The Congressional Justification provides a detailed budget request that is incorporated into the President’s Budget Proposal. It outlines NCI’s mission, goals, and objectives for the coming fiscal year and provides comparative data and analysis for the previous, current, and proposed budgets.
If an appropriations bill is not passed by the start of the fiscal year (October 1), Congress may enact a continuing resolution, which provides federal agencies with limited, short-term funding, typically at the pre-existing appropriations level. Congress may pass multiple continuing resolutions until an agreement is reached on a final budget for the full fiscal year. If a budget agreement is not reached and no continuing resolution is passed, the government faces a shutdown.
How does NCI spend its budget?
As the leader of the cancer research enterprise, collectively known as the National Cancer Program, and the largest funder of cancer research in the world, NCI manages a broad range of research, training, and information dissemination activities that reach across the entire country. NCI leadership relies on advisory committees, external scientific experts, and NCI’s subject matter experts to provide objective advice regarding scientific priorities, development of extramural program initiatives, future direction of NCI’s intramural and clinical trials programs, and the Frederick National Laboratory for Cancer Research.
There are many ways to break down NCI’s spending; the graph below shows one example. Explore the interactive NCI Budget Fact Book for additional breakdowns and details on how NCI spends its budget.
- Research project grants make up the largest portion of NCI’s budget. These are awards made to institutions or organizations to support discrete, specified projects to be performed by named investigators in areas representing their specific interest and competencies (e.g., R01, R37). In the grant-making process, NCI relies heavily on rigorous scientific peer review. Learn more about NCI’s grants policies and process.
- Centers and SPOREs include grants for NCI-Designated Cancer Centers (e.g., P20/P30 grants), as well as Specialized Programs of Research Excellence (SPOREs) that focus on a specific organ site, on a group of related cancers, or other factors.
- National Research Service Awards (NRSA) is a trainee award program that helps ensure that a diverse pool of highly trained scientists is available in appropriate scientific disciplines (e.g., T32, F30).
- Research and development (R&D) contracts include the Frederick National Laboratory for Cancer Research, the Surveillance, Epidemiology, and End Results Program (SEER), and others.
- Other research includes the following grants: career programs (K Awards), cancer education, Predoctoral to Postdoctoral Fellow Transition Awards, education projects - cooperative agreements, minority biomedical research support, as well as NCI’s grant support for extramural clinical trials networks such as the National Clinical Trials Network, the NCI Community Oncology Research Program, and NCI-supported early-phase clinical trials networks.
- Intramural research funding makes up about 18% of the NCI budget. NCI’s more than 300 intramural researchers conduct basic, pre-clinical, clinical, genomic, and population-based research.
Major areas of NCI’s work that span more than one of the categories listed above include other support for clinical trials, training and career development, and basic science. These efforts are funded through multiple mechanisms and approaches, including intramural research, extramural research, national networks, and research project grants.
How does NCI make spending decisions?
When making spending decisions, NCI leaders must balance competing priorities, commitments, and opportunities to ensure continued progress toward its mission. NCI’s external advisory committees provide objective and expert advice on coordination of the institute’s activities. The institute does not use predetermined targets for specific disease areas or research categories to make spending decisions.
In the grant-making process, NCI relies heavily on scientific peer review. The first level of review is carried out by a Scientific Review Group composed primarily of non-federal scientists who review research proposals and judge them on factors such as:
- importance of the research (significance, innovation)
- rigor and feasibility (approach)
- expertise and resources (investigator, environment)
The National Cancer Advisory Board provides the second level of review. This intensive approach, in line with NIH's peer review process, ensures that NCI supports the best science aligned with its mission.
Research proposals are also further evaluated by NCI’s scientific leaders and program staff, who consider public health significance, scientific novelty, overall representation of the research topic within the NCI portfolio, diversity of thought, and other factors.
How do NCI's funding decisions address human-based research approaches and animal studies?
In alignment with an NIH initiative announced in 2025, NCI is committed to reducing animal use in cancer research and expanding innovative, human-based science. Where scientifically valid and justified, funding opportunities may place special emphasis on these human-based approaches.
Although animal studies have played an important role in advancing biomedical science, emerging human-relevant technologies—such as organoids, computational models, and real-world data integration—offer powerful new tools that can complement or even replace animal models.
Human-based studies are particularly valuable in cancer research because individual cancers can look and behave differently based on a person's genetics, environmental exposures, individual tumor microenvironments, and immune responses. As a result, human-based science can provide more accurate insights into tumor biology and treatment response and safety, improving the likelihood that discoveries will translate into effective therapies for people with cancer.
While funding opportunities may highlight human-based research approaches, NCI will continue to fund research using laboratory animals when it is scientifically appropriate, well-justified, and conducted with proper animal welfare oversight. When animal models are proposed, applicants must clearly justify their necessity and use the minimum number of animals needed for rigorous and reproducible research.
For more information from NIH, visit How Does the NIH Initiative to Prioritize Human-Based Research Affect Research Proposing the Use of Laboratory Animals?
What is NCI’s budget history?
NCI’s overall budget appropriations are presented below for the last 20 years. The enacted appropriation levels do not include potential adjustments such as rescissions, sequestrations, supplemental funding, or Secretary's transfers that may have impacted the amount available for NCI expenditure.
Fiscal years 2017 through 2023 include funding appropriated through the 21st Century Cures Act, which authorized $1.8 billion in funding for the Cancer Moonshot over seven years. NCI’s base appropriations include funding for the Childhood Cancer Data Initiative (CCDI), $50 million per year, proposed for 10 years starting in 2020, and $28 million for the Childhood Cancer Survivorship, Treatment, Access, and Research (STAR) Act of 2018.